From: Matthew
Re: Organic Chemistry GAMSAT – Free Practice
Organic Chemistry GAMSAT Questions
Unit 2
Question 1-3
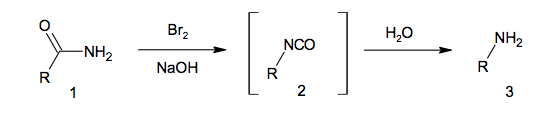
The Hoffmann rearrangement of amides 1 is traditionally carried out using bromine and sodium hydroxide in water. The resulting isocyanate 2 then hydrolyses affording primary amines 3, as depicted in the scheme below.
1. What is the by-product of the hydrolysis reaction?
A. Ethanol.
B. Water.
C. Molecular oxygen.
D. Carbon dioxide.
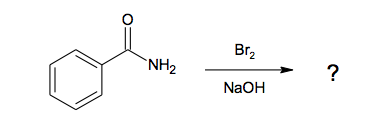
2. What is the product of the reaction below?
A. Benzoic acid.
B. Benzyl amide.
C. Aniline.
D. Benzene.
3. What is the product of the reaction below?
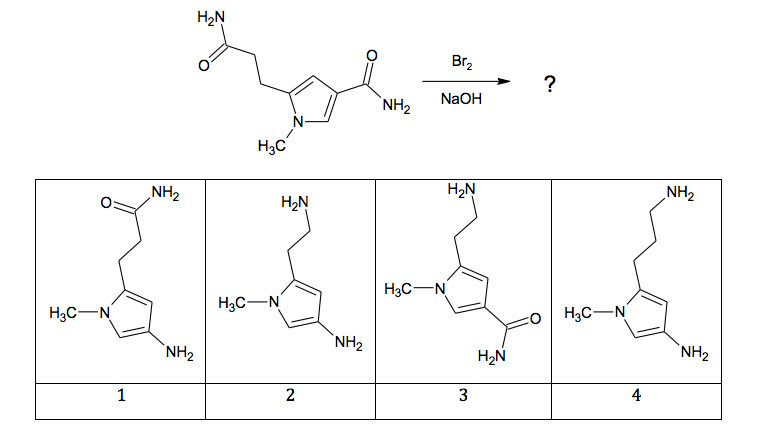
B. 2
C. 3
D. 4
Organic Chemistry GAMSAT Answers
Answer to Question 1
Isocyanates 2 (RNCO) hydrolyse through the intermediacy of carbamic acids (RNHCOOH). These unstable compounds decarboxylate (or in other words lose carbon dioxide) to afford primary amines 3. The key is D.
Answer to Question 2
The reaction affords a primary amine (aniline or phenylamine) and not a hydrocarbon compound, an acid or an amide. The key is C.
Answer to Question 3
The reaction affects both the present amide functionalities and hence structures 1 and 3 represent the wrong products. Structure 4, on the other hand, is wrong as the connectivity is incorrect (one carbon too many). The key is B.
__